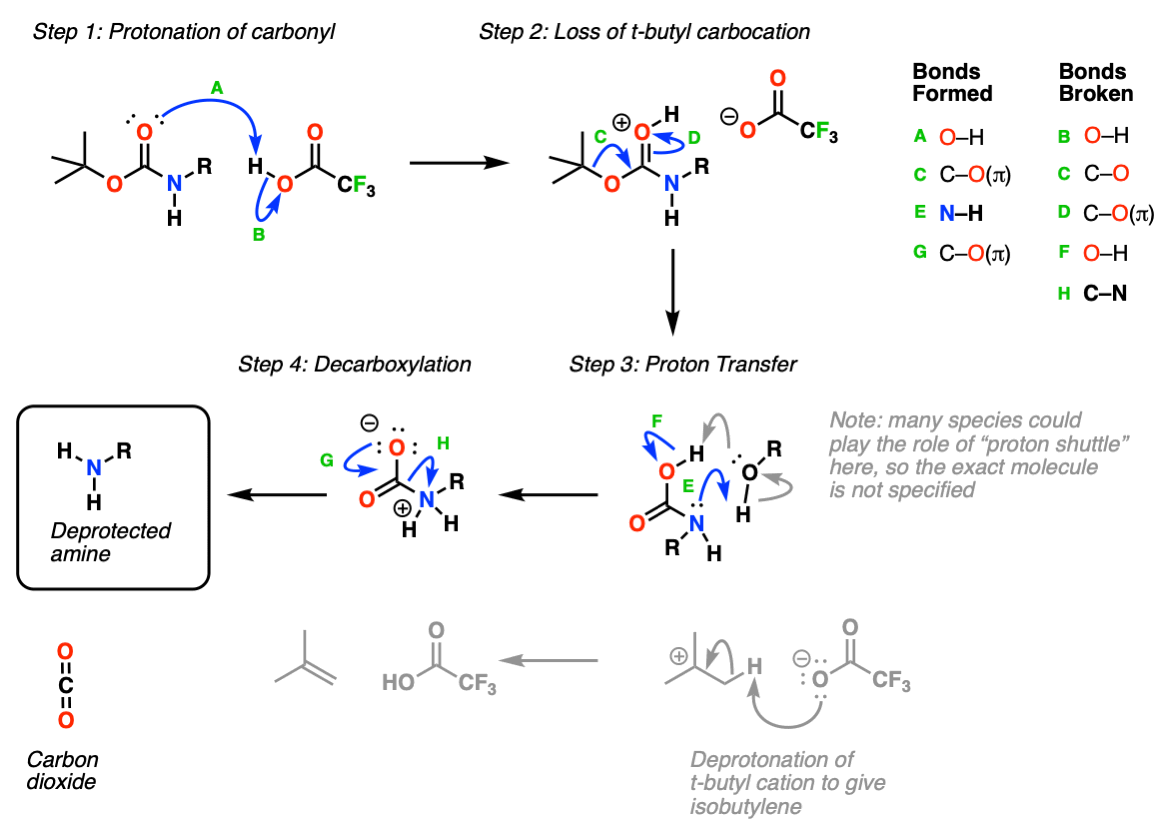
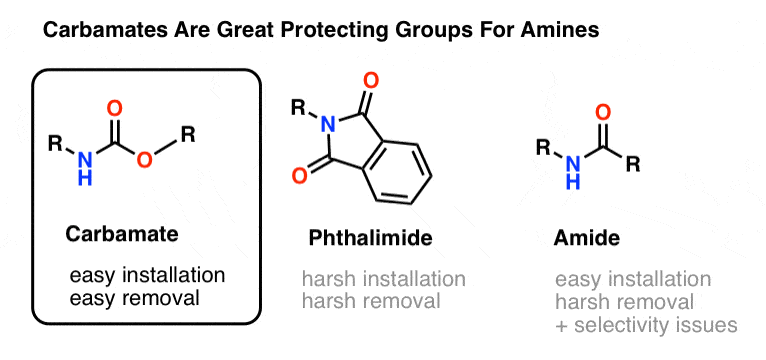
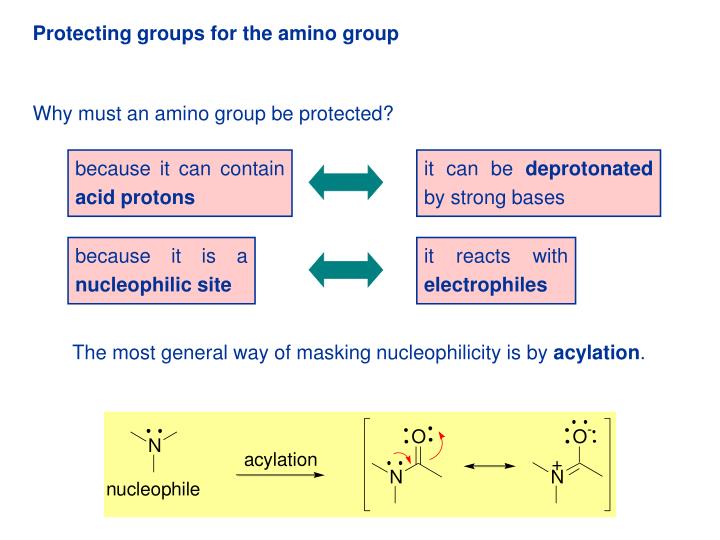
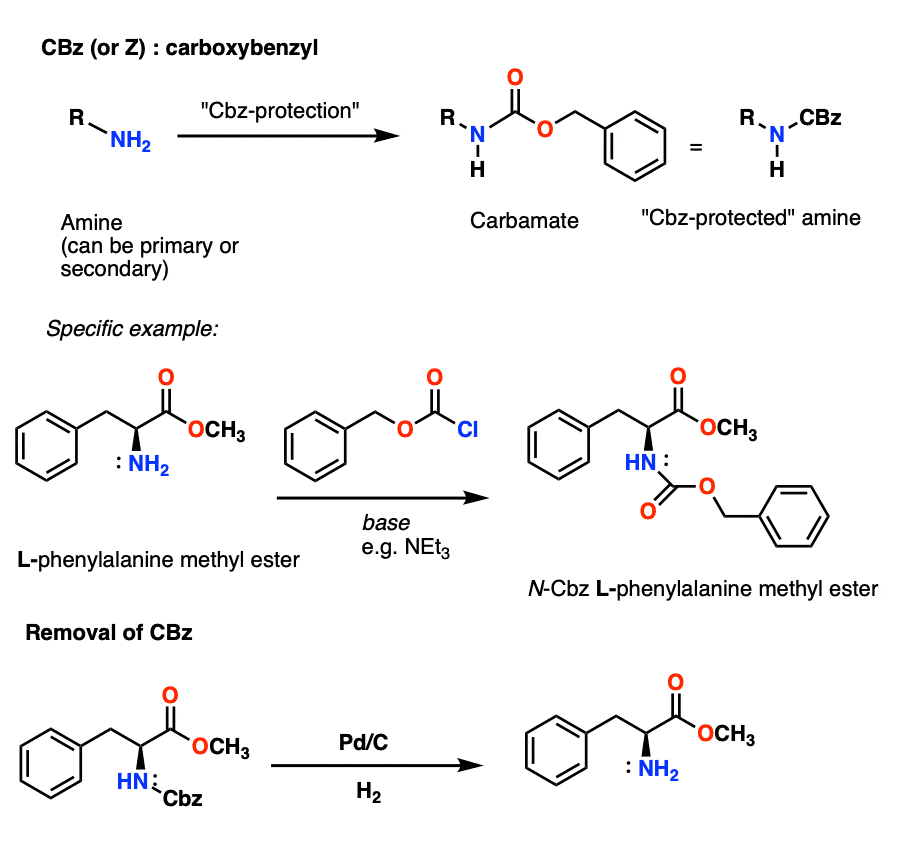
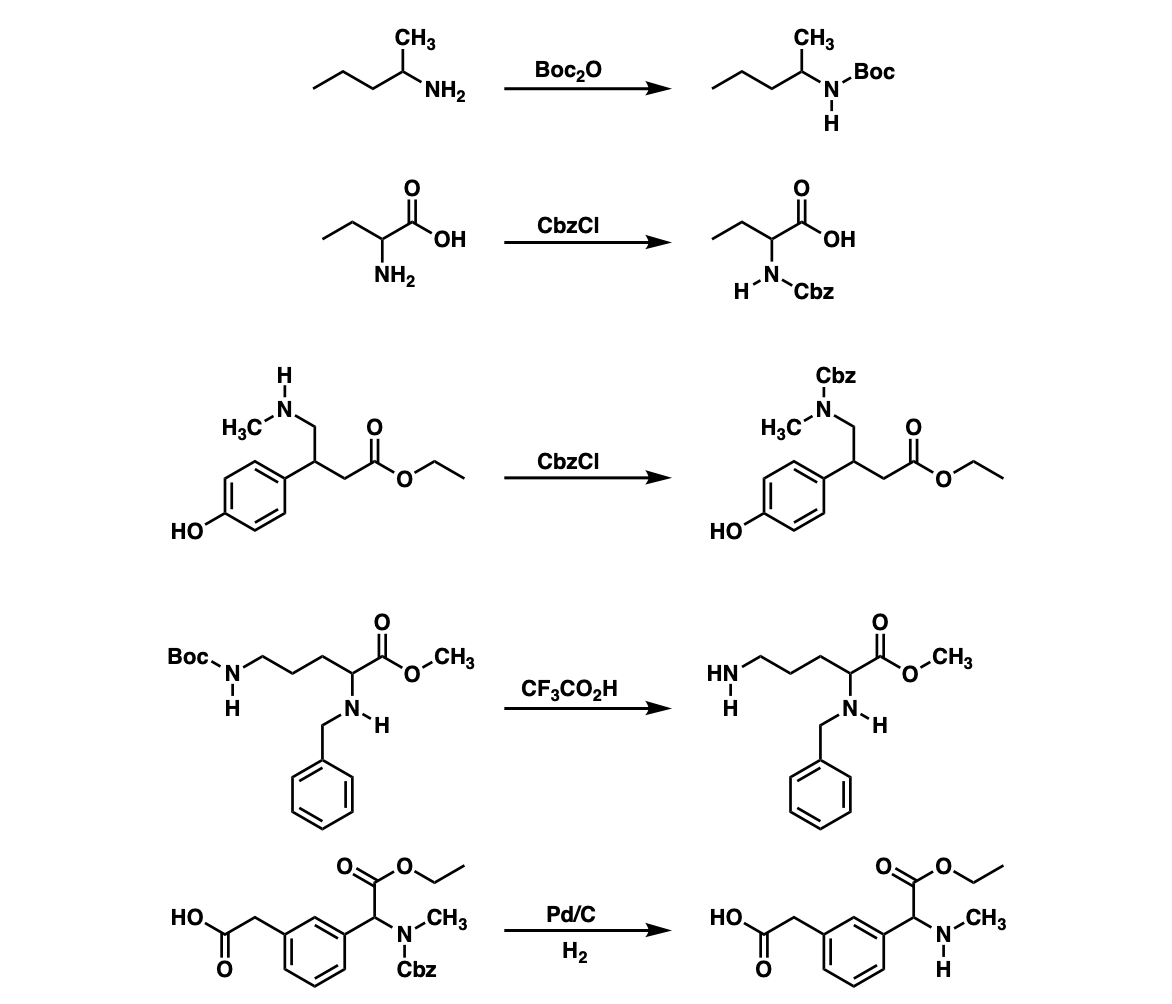
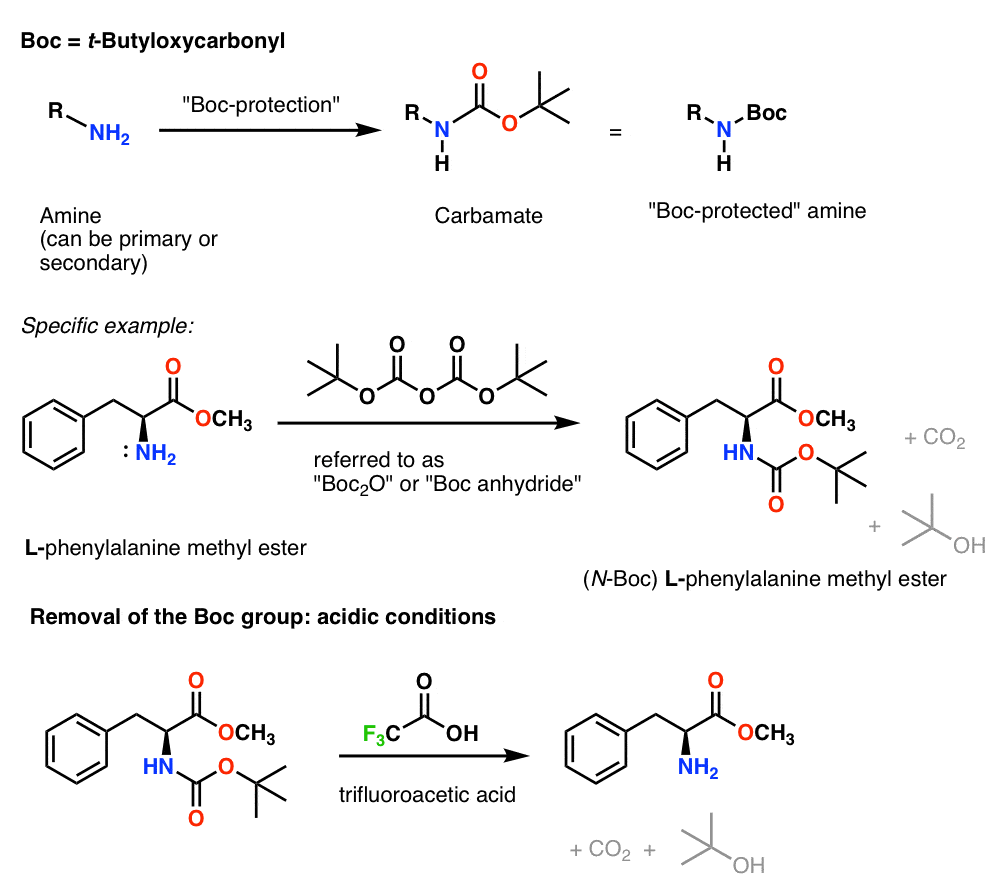
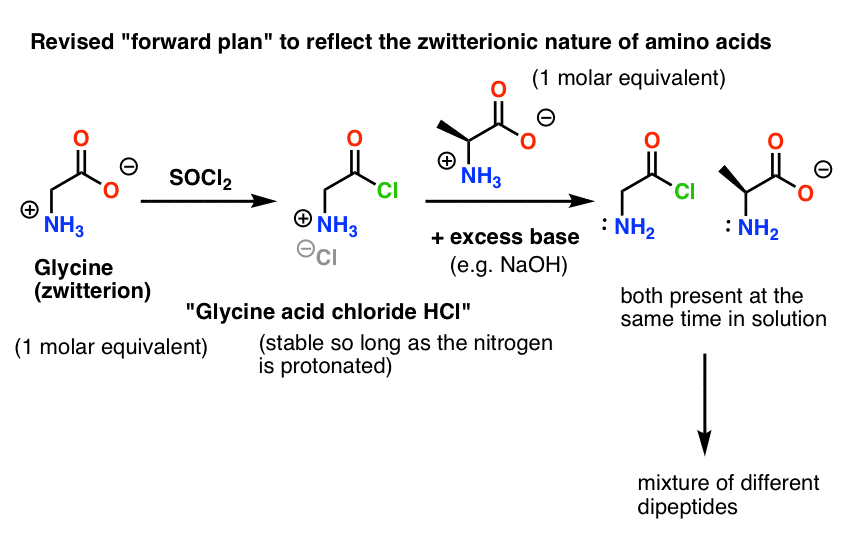
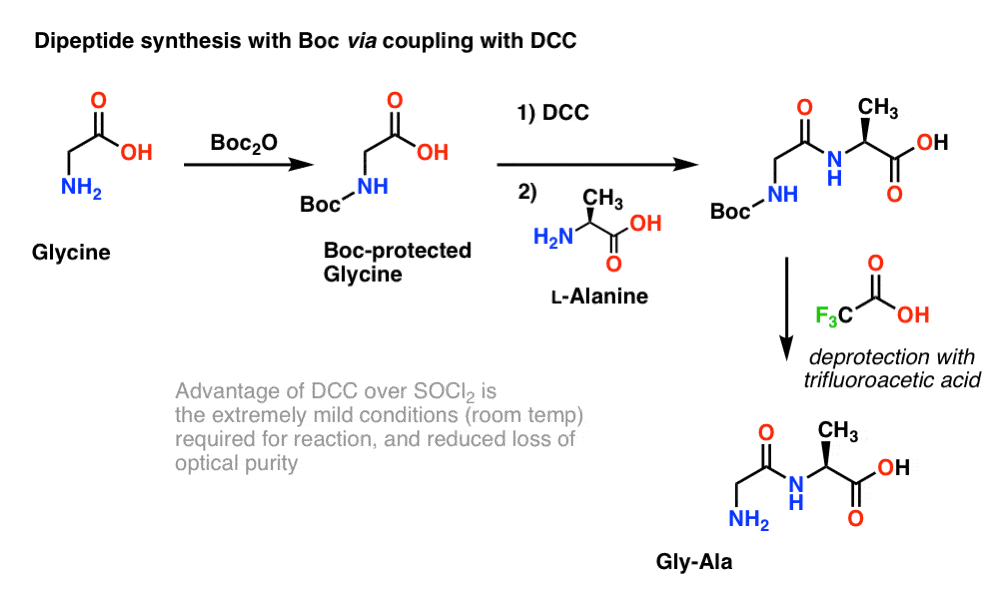
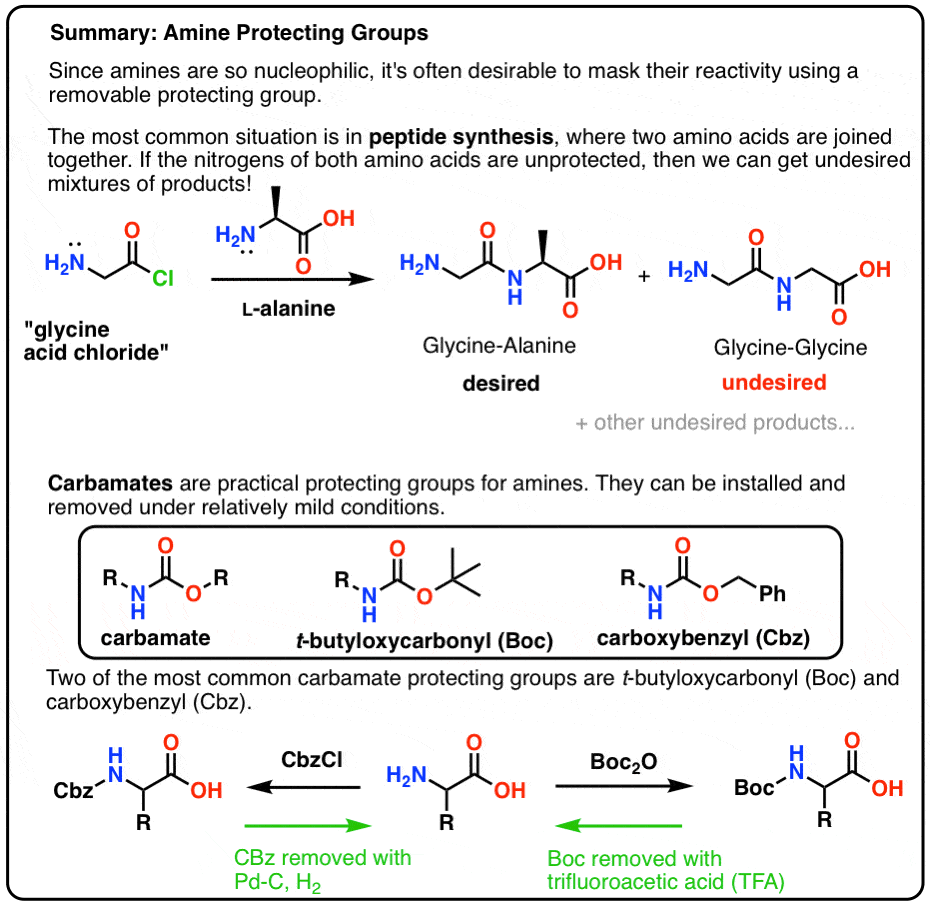
Unlocking the Potential of Phenacyl Protecting Groups: CO2-Based Formation and Photocatalytic Release of Caged Amines | The Journal of Organic Chemistry

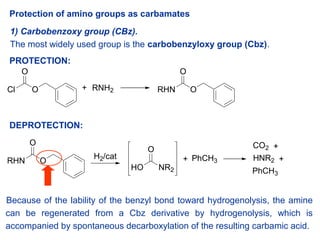
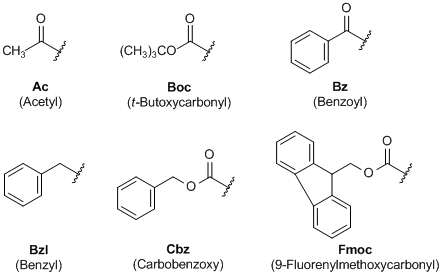
Chemical structure and short names of TFA-labile protecting groups used... | Download Scientific Diagram
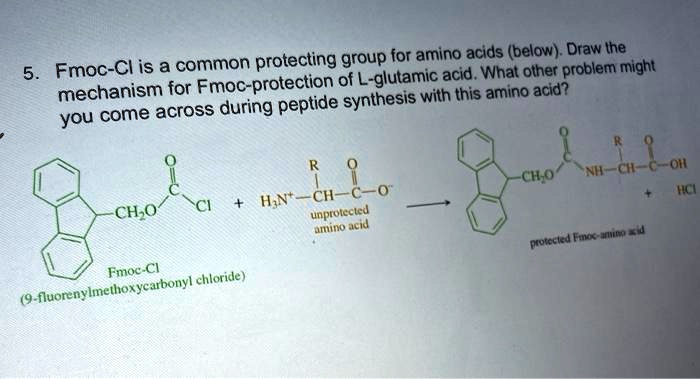
SOLVED: Fmoc-Cl is a common protecting group for amino acids. What other problem might arise with this amino acid during peptide synthesis?

Utilization of Fukuyama's sulfonamide protecting group for the synthesis of N-substituted α-amino acids and derivatives - ScienceDirect