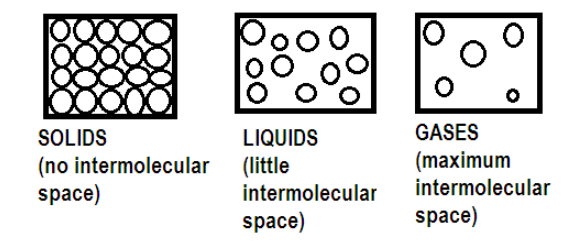
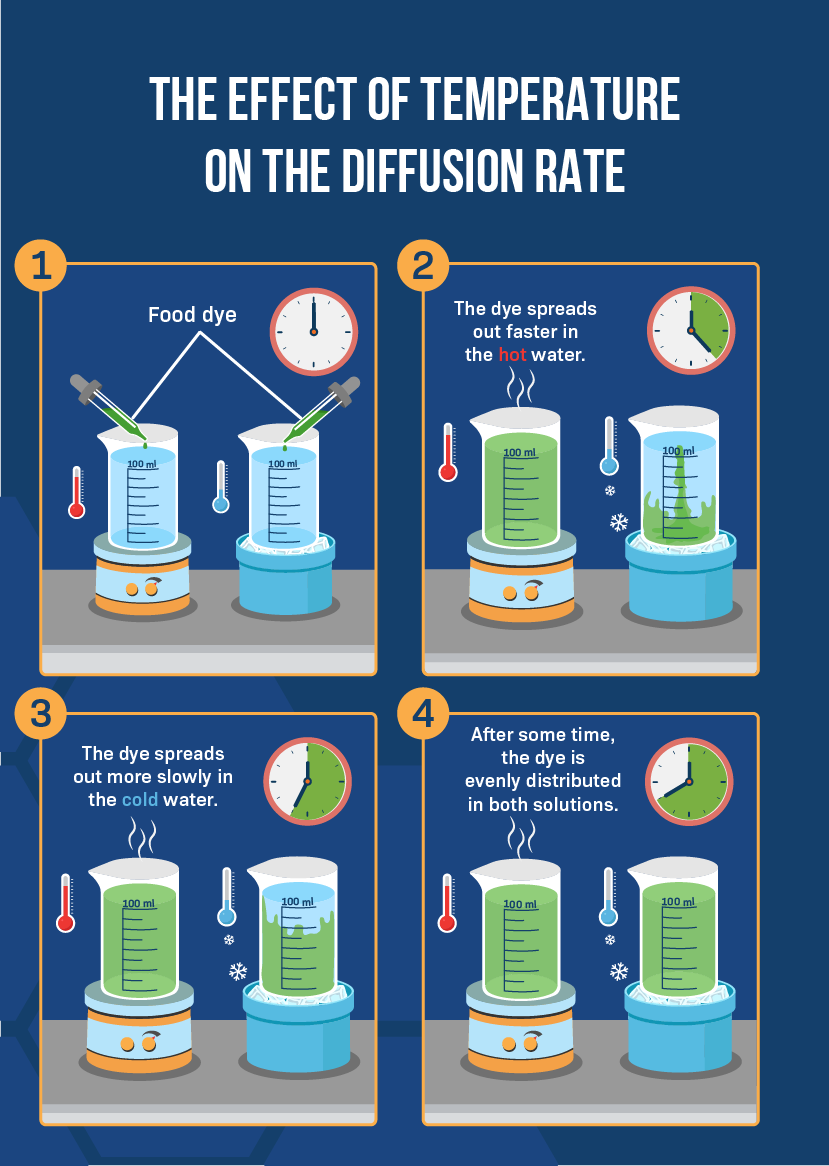
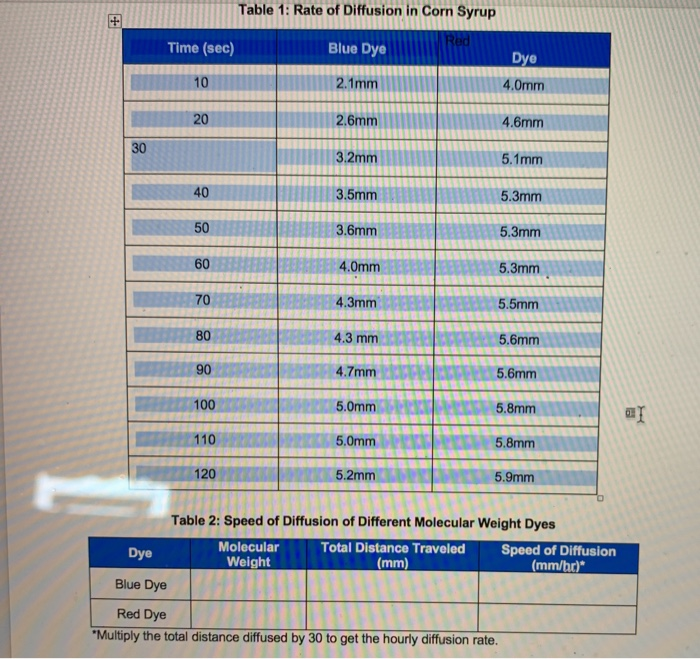
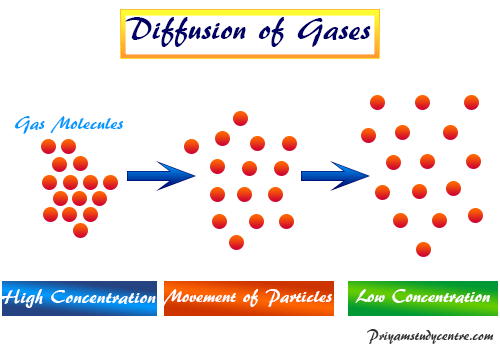
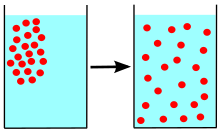
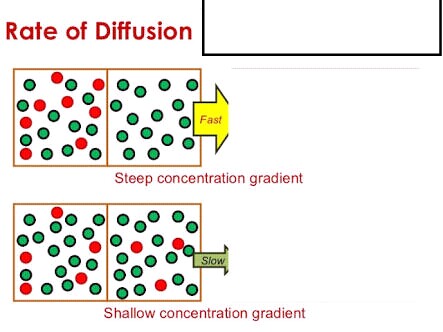
Give a simple activity/experiment to show that the rate of diffusion increases with rising temperature.

acid base - What is the relative rate of diffusion of ammonia to hydrogen chloride, both in gaseous states? - Chemistry Stack Exchange

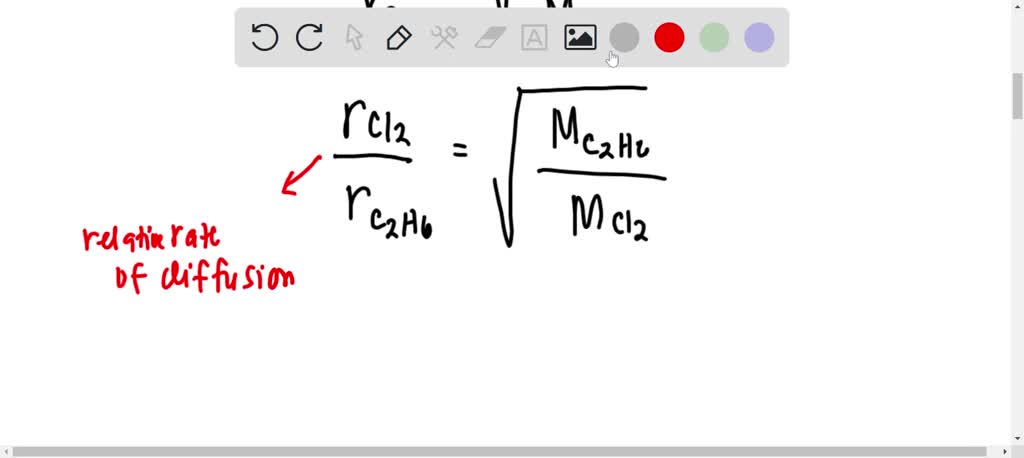
Rate of diffusion of gas is :directly proportional to its molecular massdirectly proportional to square of its molecular massinversely proportional to the square root of its moleculer massdirectly proportional to its vapour